The research team led by Associate Professor KOH Ming Joo from the NUS Department of Chemistry has developed a boron-catalysed method to transform oxetanes which are small four-membered ring molecules into larger, medicinally relevant 1,3-oxazinanes by the selective insertion of two building blocks, a carbon unit and a nitrogen unit. The research breakthrough was published in the scientific journal Nature Synthesis.
Streamlined strategy to build 1,3-oxazinanes
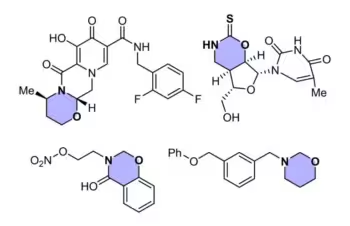
Multi-heteroatom rings such as 1,3-oxazinanes occupy a unique and important position in heterocyclic chemistry, and are frequently leveraged in drug development as local anaesthetics or for treating illnesses such as acquired immune deficiency syndrome (AIDS) and tuberculosis. However, traditional ways of preparing these six-membered heterocycles often require many separate steps, starting from specially prepared materials. This can create more chemical waste, take longer, and limit the range of structures that chemists can easily explore.
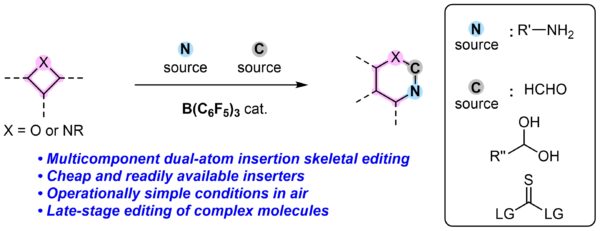
“Frustrated Lewis pair” (FLP)-driven multicomponent editing with a boron catalyst to introduce carbon and nitrogen building blocks into oxetanes. [Credit: Nature Synthesis]
Associate Professor Koh said, “We wanted to design a novel approach for the rapid assembly of 1,3-oxazinanes from cheap and readily accessible starting materials, without relying on specialised functionalities to facilitate the reaction.”
A boron-catalysed “FLP” route
The researchers developed an innovative strategy that harnesses boron-based “frustrated Lewis pair” (FLP) activation to upgrade cheap and readily available oxetanes to 1,3-oxazinanes. “We believe that FLP-mediated chemistry would provide a general way to access 1,3-oxazinanes and accelerate the synthesis of target molecules, by enabling chemoselective late-stage editing and minimising protecting group chemistry,” added Associate Professor Koh. Read the full article here.
